In this thread, I'll be detailing all the techniques I use to manufacture metal parts, as well as the following topics:
- soldering metal (what I use, how I do it, and subsequent processing)
- blackening (what I use, how, and how to achieve a natural look for cast iron and forged steel)
- electroplating or electrochemical copper plating.
*Take a few minutes to read this, and you'll be amazed, I guarantee.
So, what is electroplating? If you think it's complicated and not for you, after reading this post, you'll realize it's much simpler than you thought.
Laziness is the engine of progress. I've always looked for a way to do things faster and with less effort. So, I searched for a way to produce identical parts in large quantities. And the only way is 3D printing.
But many might argue that this isn't as authentic, that painted plastic will never look like blackened metal... and I completely agree!
But what if you coat the plastic with metal?! And then blacken it... and voila – electroplating is your thing!
So, what is electroplating? It's a method of coating one metal with another, in very simple terms. This can be used for gold plating, silver plating, zinc plating, and many other things, but we're interested in copper plating.
But! I just said it's coating another metal with metal, but what does plastic have to do with it? So, for copper plating to work, our plastic part needs to be electrically conductive. But how can we do that? One option is graphite! Regular graphite, the kind you find in pencils. But applying this powder is problematic, so there are ready-made sprays that can be used to apply a layer of graphite like a coat of spray paint.
And now our part is ready for copper plating. And if anyone thinks this is just for show and that such a part won't be able to withstand the load, it all depends on the time it takes to copper plating. The longer the process, the thicker the copper layer, and such a part can even be sanded. Next, I'll show you my experiments with loads on such parts (rims and hinges), and you'll see... no, you'll even be surprised at how durable they are!
So what have we come up with? If a part looks like metal (which it essentially is), if it bends, polishes, and blackens like metal, then what difference does it make what's inside? Many people copper-plated tin (chemically or galvanically), which is a brittle and very flexible metal. How is it any better than plastic, which is much stronger (for its intended purpose). By the way, weight (for those who prefer metal) can sometimes play a cruel joke, when a heavy cannon is torn from its carriage on a finished model during transportation due to shaking...
So, if you've read this far, I suggest you take a look at my experiments in electroforming. I'll try to explain everything in detail so that even a child can follow it. Let's go!
But first, here are some recommendations for mandatory implementation.
Protective equipment: mask, gloves, glasses.


1. Details. I glued the details made of SLA photopolymer plastic (which I printed on a 3D printer) to the CA. They need to be positioned so they don't block access to each other's side walls or touch each other. This will prevent the graphite coating from being applied properly. It's important to understand that the fasteners should be located where the part won't be visible. Large parts are best fastened at several points, preferably hidden from view and on protruding areas. I'll talk more about fasteners later. Also, closed parts (bells, buckets) need to be fastened sideways to prevent air bubbles from forming inside. This is important!

2. Graphite. Applying graphite varnish to the parts and to the adjacent wire is essential. The aerosol spray is applied like paint and is odorless, but its downside is that it's not cheap. Apply it carefully to avoid any gaps or drips. It's best to apply it once, then check everything and repeat. The layer is very thin, so it's best to apply a little more to avoid creating gaps later, but don't go overboard (again, it's not cheap, costing 17-20 euros).

3. The graphite layer is quite fragile. Don't place the part on anything rough. The holder should be designed so that the parts are always suspended and don't touch anything! You can lightly polish the surface with a very soft cloth before immersion.
Very important! You must wait until the surface is completely dry. Although graphite appears to dry within a few minutes, this is deceptive. Complete drying takes 2-4 hours! If this is ignored, the layer will be loose and fragile, the process will be slow, and a "sugar" will form on the surface.
Caution! Graphite is very finely dispersed and must be sprayed in an isolated area. If you get it on the floor, the dust will spread everywhere and be difficult to clean. You have been warned!

4. Electrical equipment. We need a DC power source. Any spare charger will do. I used a 5-volt, 1.2-amp one. This is more than enough for our purposes. We connect the positive and negative terminals of this charger to the XY-SK35H step-down converter (easy to buy on AliExpress for 10-15 euros). We'll set the desired current and volts on it (more on this device later). From this step-down converter, we need to connect two positive terminals and one negative terminal (or two negative terminals, too).

5. Anode (+). Two copper plates (pre-polished to remove oxide film with steel wool or sandpaper, important!). It's advisable to make covers out of non-woven material, like a coffee filter. This is necessary to prevent copper debris from getting into the solution (see below for the importance of this). The plates should be above the solution level, and the contacts with the wire should be on the surface.

6. It's best to secure the plates with clothespins for stability. They should be positioned so that they are directly opposite each other. The container should be larger than the volume of the solution. For 1 liter of solution, use a 1.5 liter container.
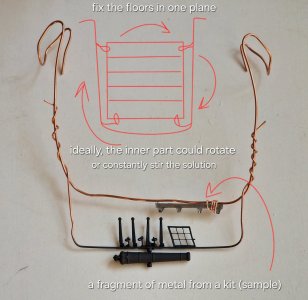
7. The details should be arranged in layers but in the same plane! Nothing should be loose or protrude beyond the main plane (very close to the plates). It's advisable to make sure the entire plane can rotate or periodically stir the solution. Ideally, connect an automatic stirrer or an aquarium pump.
Again, the sedimentation rate will slow if the solution isn't stirred. The copper will settle to the bottom, and the process will take a long time. To ensure the copper is distributed evenly throughout the solution, the solution must be constantly moving. Either stir it regularly or install an automatic system to stir it for you.

8. Cathode (-). The negative terminal must be connected to the wire with our components. The distance from the copper plates must be the same for all components. The components should never touch the copper plates. (You can make two negative terminals, and run another one to the other side; that's what I did.)

9. Solution. I bought a ready-made one, which I recommend. It's an acidic solution for bright copper plating. You can try making it yourself, but you'll need pure copper sulfate (not agricultural grade), sulfuric acid (battery grade), alcohol, salt, and thiocarbamide. But if you buy everything, the cost will be more than a liter of the ready-made solution. Yes, you'll end up with five times more raw materials, but why bother? This solution has no expiration date, and with filtering, one liter is enough for many models. Look for it in specialized stores where jewelers shop; it costs 5 euros.

10. Converter settings. This type of equipment is quite expensive if it is professional. I bought this device for 12 euros. Look for the XY-SK35H on AliExpress. How to set it up: 1) connect to the network (+- wires from the power supply), 2) turn on the Power button (ON), 3) then press SW and make sure that "IN" is NOT there, that is, so that we see the outgoing settings and not the incoming ones, 4) press V / A and in the set cv (00.00) setting, set 02.00 Volts (this parameter is not mandatory, it will automatically change as needed depending on the Ampere, but the upper limit can be set to 1-2 V), 5) press V / A again and in the set cc (0.000) setting, set 0.080 Amperes! This is important, not 0.8 but 0.08!! This parameter is the most important. 6) after inactivity, the settings will be saved. Done.
These parameters aren't universal, but rather an example with this number of parts!
You need to calculate according to the following principle:
- Start and until complete copper plating - 0.01-(0.02) Amps per square centimeter.
- After the entire surface is coated - 0.02-(0.03) Amps per cm². (But you don't have to change this if you're not in a hurry).
How to calculate the area of the detals: Take the part and draw the outlines of all the surfaces that will be copper-plated around it on a piece of paper. If the part is a cylinder, mentally unfold it into a single plane. Add up the area in square centimeters. For me, it was approximately 2 x 4 = 8 cm, 8 x 0.01 = 0.08 Amps. This step is very important for selecting the current.

11. After an hour, you can slightly increase the current (amperes. Volts are set automatically; if there are more parts, you can set the maximum to 2 volts). This isn't necessary if you're not in a hurry, but since I added a coin and a pendant to the copper plating, I increased it to 0.1 amps. After the entire surface of the parts is copper-plated, you can increase the current to 0.02-(0,03) amps per cm², but this isn't necessary.
It's important to understand that: 1) if the current (amperes) is low, the process may proceed very slowly or even stop. 2) if the current is high, the copper plating rate will be higher, but there's a risk of localized thickening and pimples. The current should be optimal, closer to the values I listed above. If the part is very complex and its area is difficult to calculate, increase the current by 5-10% every half hour and monitor the progress. If it speeds up and there are no artifacts, repeat, but do not increase it too quickly or too much and check constantly, especially during the first attempts.

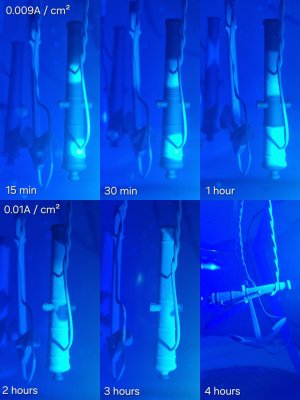
12. Process. The gun was copper-plated for over 6 hours, and the small parts for up to 5 hours. In the photo above, you can see how the copper gradually crept up onto the parts, covering them. However, one hinge (3rd from the left) and one middle rim didn't even begin to coat for almost 3 hours... I was beginning to think there was a gap in the graphite coating, but after 3 hours, these parts finally started to coat. Apparently, the copper bypassed the gap, or other parts absorbed all the current. So don't panic if you don't see anything. Hold a flashlight to the wall, and you'll see the copper growing.


13. This is the result after 6 hours of crawling and +14 hours of subsequent copper plating.
Unfortunately, I forgot to rotate the coin in the holder, and as the character in "Pirates of the Caribbean" said, the coin became part of the ship. It was firmly attached to the holder. To prevent this, you need to rotate it every couple of hours.
This result could have been achieved faster, but I decided to try the slower method to achieve the most even layer. Now, we'll speed things up.


14. Filter. Here's a clear example of why a filter is needed. After use, the plates should be rinsed with running water and the filter washed. Before the next use, the plates should be treated with steel wool.

15. Layer thickness. Depending on the copper plating time (in my case, it was 6 hours of layer growth and +24 hours of layer buildup), you can build up the thickness of a sheet of paper—0.1 mm or 100 microns. While this may seem like very little, this thickness isn't really necessary. 50-60 microns is sufficient to safely grind and polish the part. And at a thickness of 60-100 microns, the part's strength is simply incredible, but the burrs increase and the geometry changes. More on this later.

16. I forgot to remove the copper from the water after rinsing, and it turned out like this. The colors are simply stunning...

But that's not a problem. It's just oxide, and it can be easily removed with steel wool. But there's a catch, and I'll explain it in detail below!

17. Artifacts. Waves or burrs, as I understand it, appear more often on the top, so it's better to place the part upside down. To prevent this, stir the solution or rotate the part regularly. Pimples or growths (like coral) are caused by high current. (I try to avoid this. It's better to go slower, but then there won't be any extra work when sanding.) But even if these appear, copper sands perfectly with sandpaper and polishes with steel wool (but I'll explain more about polishing below!)

In this way, as you can see, you can copper plate any metal, from jewelry to coins.

For comparison, the original 1 hryvnia coin is on the left, and the copper-plated one is on the right. If you'd like, I'll coat it with a green patina to achieve an antique look; more on that later.
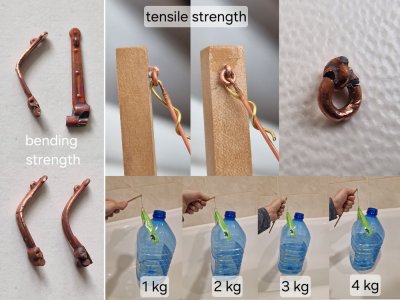
18. Strength. I tested parts with a 0.1mm layer thickness. They were so stiff in bending that they couldn't be compared to regular copper. I could almost reach a 90-degree angle before the part broke. But bending at 50-70 degrees was completely straightforward. I tested the tear-off strength of a rim of similar thickness. It's a shame I didn't have one with the ring raised, so I could have tested it more accurately. Even so, it's clear that the rim easily withstood over 4 kilograms and broke at 5. I'm sure it would have withstood even more if it hadn't bent from a "flat" position. But then, the glued joint might not have held up. It could have simply torn away from the hole (wood or glue). But even if we take this figure into account, I'm not sure most of you have a model that weighs even as much as this rim alone. Therefore, I think, for those who want strength (although I am sure that half will be more than enough) - you can safely build up 100 microns, but there is one But!

19. Geometry. Due to the increased layer thickness, especially on small parts, changes in geometry will be visible. Edges will thicken and round, and textures will become smoother.
In the photo: on the left is a 0.4 mm thick plastic part. In the center is a 50 micron copper layer, on the right is a 100 micron layer. (*Painted plastic rim).
If strength and thickness are required, then this should be calculated as much as possible during modeling and taken into account when constructing the geometry; this will avoid problems. If the layer thickness is not too thick and extreme strength is not required, a 30-40 micron layer can be used - that's a third of a sheet of paper - which, with ideal geometry and minimal sanding, is more than sufficient.
The geometry of parts can be refined experimentally. In a 3D program, you can model dozens of variations in size and shape in just a few minutes. Print hundreds of parts at once and then copper-plated them in the same quantity, then choose the best ones. You can make them slightly uneven, with different positions, to achieve maximum realism... but it's impossible to manually produce the same number in such a short time using the standard method, that's a fact.
I'm already preparing a second set of tests to demonstrate different conditions, how to and how not to copper-plated them. I also plan to test copper varnish (if I buy it). It's even more expensive and harder to find than graphite. But it works much faster, is more predictable, and the copper grows not from the holders, but over the entire surface.
Naturally, I also plan to show you how to blacken it all. This process also involves some tricks, as the cannons need to be blackened using one method to make them look like cast iron. And everything else - anchors, rims, hinges - should be blackened using a different method to make them look like forged steel.
To be continued.
- soldering metal (what I use, how I do it, and subsequent processing)
- blackening (what I use, how, and how to achieve a natural look for cast iron and forged steel)
- electroplating or electrochemical copper plating.
*Take a few minutes to read this, and you'll be amazed, I guarantee.
So, what is electroplating? If you think it's complicated and not for you, after reading this post, you'll realize it's much simpler than you thought.
Laziness is the engine of progress. I've always looked for a way to do things faster and with less effort. So, I searched for a way to produce identical parts in large quantities. And the only way is 3D printing.
But many might argue that this isn't as authentic, that painted plastic will never look like blackened metal... and I completely agree!
But what if you coat the plastic with metal?! And then blacken it... and voila – electroplating is your thing!
So, what is electroplating? It's a method of coating one metal with another, in very simple terms. This can be used for gold plating, silver plating, zinc plating, and many other things, but we're interested in copper plating.
But! I just said it's coating another metal with metal, but what does plastic have to do with it? So, for copper plating to work, our plastic part needs to be electrically conductive. But how can we do that? One option is graphite! Regular graphite, the kind you find in pencils. But applying this powder is problematic, so there are ready-made sprays that can be used to apply a layer of graphite like a coat of spray paint.
And now our part is ready for copper plating. And if anyone thinks this is just for show and that such a part won't be able to withstand the load, it all depends on the time it takes to copper plating. The longer the process, the thicker the copper layer, and such a part can even be sanded. Next, I'll show you my experiments with loads on such parts (rims and hinges), and you'll see... no, you'll even be surprised at how durable they are!
So what have we come up with? If a part looks like metal (which it essentially is), if it bends, polishes, and blackens like metal, then what difference does it make what's inside? Many people copper-plated tin (chemically or galvanically), which is a brittle and very flexible metal. How is it any better than plastic, which is much stronger (for its intended purpose). By the way, weight (for those who prefer metal) can sometimes play a cruel joke, when a heavy cannon is torn from its carriage on a finished model during transportation due to shaking...
So, if you've read this far, I suggest you take a look at my experiments in electroforming. I'll try to explain everything in detail so that even a child can follow it. Let's go!
But first, here are some recommendations for mandatory implementation.
Protective equipment: mask, gloves, glasses.


1. Details. I glued the details made of SLA photopolymer plastic (which I printed on a 3D printer) to the CA. They need to be positioned so they don't block access to each other's side walls or touch each other. This will prevent the graphite coating from being applied properly. It's important to understand that the fasteners should be located where the part won't be visible. Large parts are best fastened at several points, preferably hidden from view and on protruding areas. I'll talk more about fasteners later. Also, closed parts (bells, buckets) need to be fastened sideways to prevent air bubbles from forming inside. This is important!

2. Graphite. Applying graphite varnish to the parts and to the adjacent wire is essential. The aerosol spray is applied like paint and is odorless, but its downside is that it's not cheap. Apply it carefully to avoid any gaps or drips. It's best to apply it once, then check everything and repeat. The layer is very thin, so it's best to apply a little more to avoid creating gaps later, but don't go overboard (again, it's not cheap, costing 17-20 euros).

3. The graphite layer is quite fragile. Don't place the part on anything rough. The holder should be designed so that the parts are always suspended and don't touch anything! You can lightly polish the surface with a very soft cloth before immersion.
Very important! You must wait until the surface is completely dry. Although graphite appears to dry within a few minutes, this is deceptive. Complete drying takes 2-4 hours! If this is ignored, the layer will be loose and fragile, the process will be slow, and a "sugar" will form on the surface.
Caution! Graphite is very finely dispersed and must be sprayed in an isolated area. If you get it on the floor, the dust will spread everywhere and be difficult to clean. You have been warned!

4. Electrical equipment. We need a DC power source. Any spare charger will do. I used a 5-volt, 1.2-amp one. This is more than enough for our purposes. We connect the positive and negative terminals of this charger to the XY-SK35H step-down converter (easy to buy on AliExpress for 10-15 euros). We'll set the desired current and volts on it (more on this device later). From this step-down converter, we need to connect two positive terminals and one negative terminal (or two negative terminals, too).

5. Anode (+). Two copper plates (pre-polished to remove oxide film with steel wool or sandpaper, important!). It's advisable to make covers out of non-woven material, like a coffee filter. This is necessary to prevent copper debris from getting into the solution (see below for the importance of this). The plates should be above the solution level, and the contacts with the wire should be on the surface.

6. It's best to secure the plates with clothespins for stability. They should be positioned so that they are directly opposite each other. The container should be larger than the volume of the solution. For 1 liter of solution, use a 1.5 liter container.

7. The details should be arranged in layers but in the same plane! Nothing should be loose or protrude beyond the main plane (very close to the plates). It's advisable to make sure the entire plane can rotate or periodically stir the solution. Ideally, connect an automatic stirrer or an aquarium pump.
Again, the sedimentation rate will slow if the solution isn't stirred. The copper will settle to the bottom, and the process will take a long time. To ensure the copper is distributed evenly throughout the solution, the solution must be constantly moving. Either stir it regularly or install an automatic system to stir it for you.

8. Cathode (-). The negative terminal must be connected to the wire with our components. The distance from the copper plates must be the same for all components. The components should never touch the copper plates. (You can make two negative terminals, and run another one to the other side; that's what I did.)

9. Solution. I bought a ready-made one, which I recommend. It's an acidic solution for bright copper plating. You can try making it yourself, but you'll need pure copper sulfate (not agricultural grade), sulfuric acid (battery grade), alcohol, salt, and thiocarbamide. But if you buy everything, the cost will be more than a liter of the ready-made solution. Yes, you'll end up with five times more raw materials, but why bother? This solution has no expiration date, and with filtering, one liter is enough for many models. Look for it in specialized stores where jewelers shop; it costs 5 euros.

10. Converter settings. This type of equipment is quite expensive if it is professional. I bought this device for 12 euros. Look for the XY-SK35H on AliExpress. How to set it up: 1) connect to the network (+- wires from the power supply), 2) turn on the Power button (ON), 3) then press SW and make sure that "IN" is NOT there, that is, so that we see the outgoing settings and not the incoming ones, 4) press V / A and in the set cv (00.00) setting, set 02.00 Volts (this parameter is not mandatory, it will automatically change as needed depending on the Ampere, but the upper limit can be set to 1-2 V), 5) press V / A again and in the set cc (0.000) setting, set 0.080 Amperes! This is important, not 0.8 but 0.08!! This parameter is the most important. 6) after inactivity, the settings will be saved. Done.
These parameters aren't universal, but rather an example with this number of parts!
You need to calculate according to the following principle:
- Start and until complete copper plating - 0.01-(0.02) Amps per square centimeter.
- After the entire surface is coated - 0.02-(0.03) Amps per cm². (But you don't have to change this if you're not in a hurry).
How to calculate the area of the detals: Take the part and draw the outlines of all the surfaces that will be copper-plated around it on a piece of paper. If the part is a cylinder, mentally unfold it into a single plane. Add up the area in square centimeters. For me, it was approximately 2 x 4 = 8 cm, 8 x 0.01 = 0.08 Amps. This step is very important for selecting the current.

11. After an hour, you can slightly increase the current (amperes. Volts are set automatically; if there are more parts, you can set the maximum to 2 volts). This isn't necessary if you're not in a hurry, but since I added a coin and a pendant to the copper plating, I increased it to 0.1 amps. After the entire surface of the parts is copper-plated, you can increase the current to 0.02-(0,03) amps per cm², but this isn't necessary.
It's important to understand that: 1) if the current (amperes) is low, the process may proceed very slowly or even stop. 2) if the current is high, the copper plating rate will be higher, but there's a risk of localized thickening and pimples. The current should be optimal, closer to the values I listed above. If the part is very complex and its area is difficult to calculate, increase the current by 5-10% every half hour and monitor the progress. If it speeds up and there are no artifacts, repeat, but do not increase it too quickly or too much and check constantly, especially during the first attempts.

12. Process. The gun was copper-plated for over 6 hours, and the small parts for up to 5 hours. In the photo above, you can see how the copper gradually crept up onto the parts, covering them. However, one hinge (3rd from the left) and one middle rim didn't even begin to coat for almost 3 hours... I was beginning to think there was a gap in the graphite coating, but after 3 hours, these parts finally started to coat. Apparently, the copper bypassed the gap, or other parts absorbed all the current. So don't panic if you don't see anything. Hold a flashlight to the wall, and you'll see the copper growing.


13. This is the result after 6 hours of crawling and +14 hours of subsequent copper plating.
Unfortunately, I forgot to rotate the coin in the holder, and as the character in "Pirates of the Caribbean" said, the coin became part of the ship. It was firmly attached to the holder. To prevent this, you need to rotate it every couple of hours.
This result could have been achieved faster, but I decided to try the slower method to achieve the most even layer. Now, we'll speed things up.


14. Filter. Here's a clear example of why a filter is needed. After use, the plates should be rinsed with running water and the filter washed. Before the next use, the plates should be treated with steel wool.

15. Layer thickness. Depending on the copper plating time (in my case, it was 6 hours of layer growth and +24 hours of layer buildup), you can build up the thickness of a sheet of paper—0.1 mm or 100 microns. While this may seem like very little, this thickness isn't really necessary. 50-60 microns is sufficient to safely grind and polish the part. And at a thickness of 60-100 microns, the part's strength is simply incredible, but the burrs increase and the geometry changes. More on this later.

16. I forgot to remove the copper from the water after rinsing, and it turned out like this. The colors are simply stunning...

But that's not a problem. It's just oxide, and it can be easily removed with steel wool. But there's a catch, and I'll explain it in detail below!

17. Artifacts. Waves or burrs, as I understand it, appear more often on the top, so it's better to place the part upside down. To prevent this, stir the solution or rotate the part regularly. Pimples or growths (like coral) are caused by high current. (I try to avoid this. It's better to go slower, but then there won't be any extra work when sanding.) But even if these appear, copper sands perfectly with sandpaper and polishes with steel wool (but I'll explain more about polishing below!)

In this way, as you can see, you can copper plate any metal, from jewelry to coins.

For comparison, the original 1 hryvnia coin is on the left, and the copper-plated one is on the right. If you'd like, I'll coat it with a green patina to achieve an antique look; more on that later.

18. Strength. I tested parts with a 0.1mm layer thickness. They were so stiff in bending that they couldn't be compared to regular copper. I could almost reach a 90-degree angle before the part broke. But bending at 50-70 degrees was completely straightforward. I tested the tear-off strength of a rim of similar thickness. It's a shame I didn't have one with the ring raised, so I could have tested it more accurately. Even so, it's clear that the rim easily withstood over 4 kilograms and broke at 5. I'm sure it would have withstood even more if it hadn't bent from a "flat" position. But then, the glued joint might not have held up. It could have simply torn away from the hole (wood or glue). But even if we take this figure into account, I'm not sure most of you have a model that weighs even as much as this rim alone. Therefore, I think, for those who want strength (although I am sure that half will be more than enough) - you can safely build up 100 microns, but there is one But!

19. Geometry. Due to the increased layer thickness, especially on small parts, changes in geometry will be visible. Edges will thicken and round, and textures will become smoother.
In the photo: on the left is a 0.4 mm thick plastic part. In the center is a 50 micron copper layer, on the right is a 100 micron layer. (*Painted plastic rim).
If strength and thickness are required, then this should be calculated as much as possible during modeling and taken into account when constructing the geometry; this will avoid problems. If the layer thickness is not too thick and extreme strength is not required, a 30-40 micron layer can be used - that's a third of a sheet of paper - which, with ideal geometry and minimal sanding, is more than sufficient.
The geometry of parts can be refined experimentally. In a 3D program, you can model dozens of variations in size and shape in just a few minutes. Print hundreds of parts at once and then copper-plated them in the same quantity, then choose the best ones. You can make them slightly uneven, with different positions, to achieve maximum realism... but it's impossible to manually produce the same number in such a short time using the standard method, that's a fact.
I'm already preparing a second set of tests to demonstrate different conditions, how to and how not to copper-plated them. I also plan to test copper varnish (if I buy it). It's even more expensive and harder to find than graphite. But it works much faster, is more predictable, and the copper grows not from the holders, but over the entire surface.
Naturally, I also plan to show you how to blacken it all. This process also involves some tricks, as the cannons need to be blackened using one method to make them look like cast iron. And everything else - anchors, rims, hinges - should be blackened using a different method to make them look like forged steel.
To be continued.
Last edited: